|
In simple words, serial dilution is the process of stepwise dilution of a solution with an associated dilution factor.In biology, serial dilution is often associated with reducing the concentration of cells in a culture to simplify the operation.Objectives of Serial dilution The objective of the serial dilution method is to estimate the concentration (number of organisms, bacteria, viruses, or colonies) of an unknown sample by enumeration of the number of colonies cultured from serial dilutions of the sample.

Make A 10 Fold Serial Dilution Serial Dilution IsIn serial dilution, the density of cells is reduced in each step so that it is easier to calculate the concentration of the cells in the original solution by calculating the total dilution over the entire series. Serial dilutions are commonly performed to avoid having to pipette very small volumes (1-10 l) to make a dilution of a solution. By diluting a sample in a controlled way, it is possible to obtain incubated culture plates with an easily countable number of colonies (around 30100) and calculate the number of microbes present in the sample. Serial dilution formulacalculations Serial dilution involves the process of taking a sample and diluting it through a series of standard volumes of sterile diluent, which can either be distilled water or 0.9 saline. 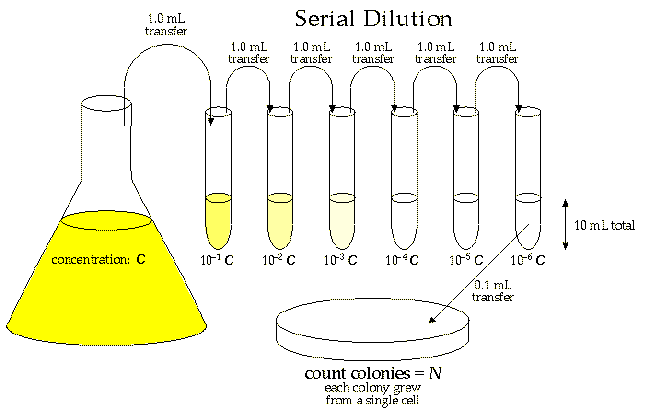
Depending on the estimated concentration of cellsorganisms in a sample, the extent of dilution is determined. For e.g., if a water sample is taken from an extremely polluted environment, the dilution factor is increased. In contrast, for a less contaminated sample, a low dilution factor might be sufficient. Serial two-fold and ten-fold dilutions are commonly used to titer antibodies or prepare diluted analytes in the laboratory. The dilution factor in a serial dilution can be determined either for an individual test tube or can be calculated as a total dilution factor in the entire series. The dilution factor of each tube in a set: For a ten-fold dilution, 1 ml of sample is added to 9 ml of diluent. In this case, the dilution factor for that test tube will be: After the first tube, each tube is the dilution of the previous dilution tube. Now, for total dilution factor, Total dilution factor for the second tube dilution of first tube dilution of the second tube. The sample is then added to the first tube to make the total volume of 10 ml. The dilution is thoroughly mixed by emptying and filling the pipette several times. The pipette tip is discarded, and a new pipette tip is attached to the pipette. Now, 1 ml of mixture is taken from the 10 -1 dilution and is emptied into the second tube. The same process is then repeated for the remaining tube, taking 1 ml from the previous tube and adding it to the next 9 ml diluents. As six tubes are used, the final dilution for the bacteriacells will be 10 -6 (1 in 1,000,000). ApplicationsUses Serial dilution is performed in a number of experimental sciences like biochemistry, pharmacology, physics, and homeopathy. Serial dilution is used in microbiology to estimate the concentration or number of cellsorganisms in a sample to obtain an incubated plate with an easily countable number of colonies. In biochemistry, serial dilution is used to obtain the desired concentration of reagents and chemicals from a higher concentration. In pharmaceutical laboratories, serial dilution is performed to receive the necessary concentration of chemicals and compounds as this method is more effective than individual dilutions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Home
- About Brooke
- Blog
- Contact
- The bear and the dragon by tom clancy
- Nepali song movie
- Super mario galaxy 2 rom pc
- Jummah khutbah pdf
- Devil jin tekken 5
- How to change language in photoshop spell check
- 3dmgame-dll missing mad max
- Kdrama the heirs
- Juicer 3 build 263
- Mutilate a doll 2 free download
- Banania poster
- Download res2dinv full crack
 RSS Feed
RSS Feed
